Published 22 October 2020 by Meeri Kim
Filming the Most Fundamental Processes in Chemistry
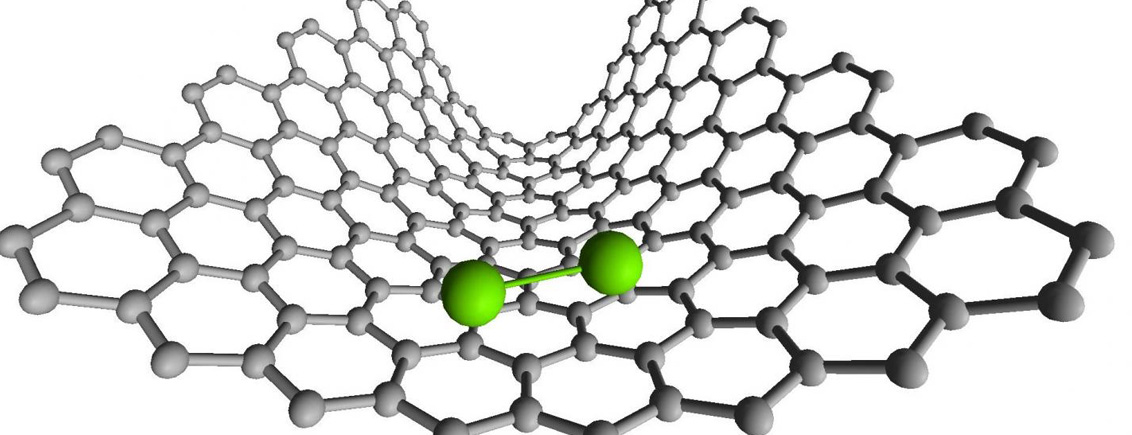
Photo/Credits: University of Nottingham
With the help of an electron microscope, two of the most fundamental processes in chemistry – chemical bonding and crystal formation – have been caught on film for the first time.
Earlier this year, a team of researchers from the United Kingdom, Germany and Japan published two studies that revealed their groundbreaking footage to the world. The first one revealed the breaking and formation of a metallic bond, while the second focused on the exact moment of crystal nucleation within three different metals. Electron microscopy makes the direct observation of these everyday chemical occurrences possible, whose mechanisms may be more complex than or even clash with those taught in school.
“Maybe some of the mechanisms of chemical reactions listed in textbooks are correct and we just need to confirm them – or maybe they’re completely different,” says Andrei Khlobystov, Professor of Nanomaterials and Director of the Nanoscale & Microscale Research Centre at the University of Nottingham. “It’s a very fundamental question, and that’s exactly what electron microscopy can help us to answer.”
Nanoscale Test Tubes
During his early career as an organic chemist, Khlobystov became interested in the imaging and manipulation of individual molecules and atoms. In particular, he was intrigued by the idea of using carbon nanotubes as test tubes to hold his tiny samples in place during imaging. Carbon nanotubes are hollow tubes made of carbon atoms with a diameter about 80,000 times thinner than a single strand of hair.
The narrow structures allowed him to carefully position atoms and molecules for transmission electron microscopy (TEM), an imaging technique that passes a beam of high-energy electrons through a sample to probe its structure. Khlobystov saw the potential of TEM for chemistry, even though at that point, it was used mainly for research in physics, materials science and engineering. Chemists, for the most part, still employ spectroscopy instead of imaging methods.
“Normally, if you think about how we study bonds breaking, we use spectroscopy. There are some excellent spectroscopy methods that are very fast and unfold this process step by step, which is wonderful,” he explains. “But the problem with spectroscopy is that you have many atoms and molecules contributing to the signal.”
TEM, in combination with carbon nanotubes, lets researchers zoom in on the actions of an individual atom or molecule in real time with atomic resolution.
Dual-Use Electron Beam
Khlobystov and his colleagues took the idea of atomic imaging with TEM a step further when they set out to watch processes like chemical bonding and crystal formation unfold. For both studies, the electron beam served as an imaging probe as well as a stimulus to kick-start the processes.
In the chemical bonding study, video footage initially shows two rhenium atoms bonded together as a dirhenium molecule within the carbon nano test tube. Because electrons have mass, the electron beam of the microscope transfers enough kinetic energy to break the bond between them. The molecule is seen to vibrate while its individual atoms elongate into ellipses as the center distance between them increases. Finally, the atoms separate completely, vibration ceases, and their shapes become circular.
The two atoms move freely and independently until, a few seconds later, they recombine into a dirhenium molecule once more. By studying each frame of the video, the researchers could measure the distance between the atoms to determine the type of bonding that occurred. Transition metals like rhenium form bonds of different order, from single to quintuple bonds, and the results demonstrate that dirhenium most frequently adopts a quadruple bond order.
The second experiment used the electron beam’s energy to initiate and power the nucleation process of metal crystallites. As one of its roles, the beam delivered additional atoms to a cluster of three iron atoms on the wall of the carbon nanotube. The cluster served as the seed, and as more iron atoms were added, the structure went through various stages of nucleation until it became an ordered crystallite.
“Crystal nucleation is a very mysterious process because it occurs with a tiny number of atoms, and it’s difficult to follow in real time with atomic resolution,” says Khlobystov. “But again, because the electron microscope gives you atomic resolution and can promote the process at the same time by giving energy to the atoms from the electron beam, we discovered the atomic mechanism of crystal nucleus formation.”
Electron Microscopy for Chemistry
Beyond a better understanding of the basic mechanisms of chemistry, the results also have larger implications for improvements in material design, discovery of new materials, catalysis, and other industrial applications. They also highlight electron microscopy as a valuable tool for chemists. Both TEM studies provide new insights into common chemical processes that would have been difficult or even impossible to gather with any other technique.
While use of electron microscopy still remains rare in his field, Khlobystov hopes that continued improvements in the technology will lead to increased adoption by chemists in the future.
“Electron microscopy has advanced now to the point where chemists like myself can use it to answer chemical questions,” he thinks. “In the next 10 or 20 years, we might start seeing more electron microscopes as part of the normal chemical laboratory, like spectrometers. Maybe we’ll start using electron microscopes in the same way.”