Veröffentlicht 10. November 2017 von Judith M. Reichel
The Hungry Brain
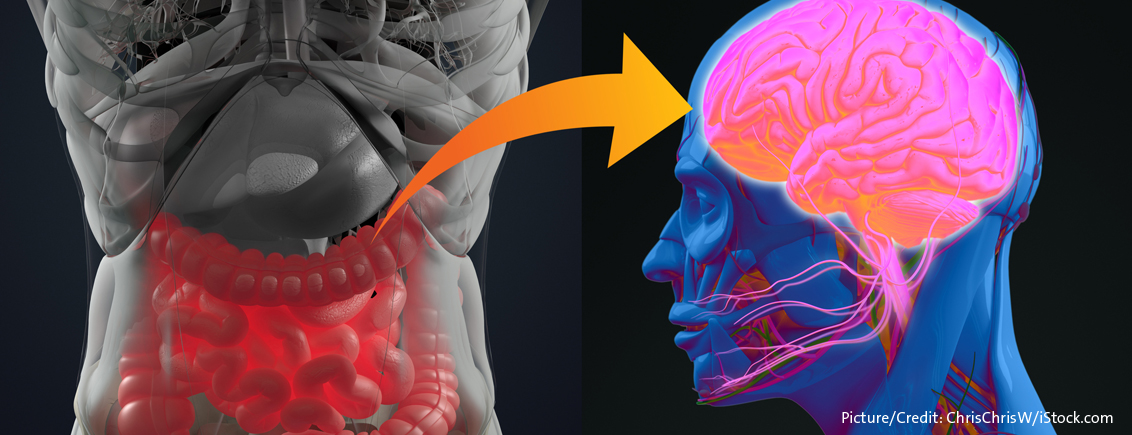
Under normal, healthy conditions we eat whenever we are feeling hungry. In addition to the feeling of hunger, we also often have an appetite for a specific kind of food, and sometimes we simply crave the pleasure a certain food like chocolate or pizza may provide us. This pleasure is part of the hedonic aspect of food and eating. In fact, anhedonia or the absence of experiencing pleasure from previously pleasurable activities, such as eating enjoyable food, is a hallmark of depression. The hedonic feeling originates from the pleasure centre of the brain, which is the same one that lights up when addicts ‚get a fix‘. Hedonic eating occurs independently of the gut-brain axis, which is why you will keep eating those crisps and chocolate, even when you know, you’re full. Hence, sayings like “These chips are addictive!” are much closer to the biological truth than many realise.
But how do we know that we are hungry? Being aware of your surrounding and/or your internal feelings is the definition of consciousness. And a major hub for consciousness is a very primal brain structure, called the thalamus. This structure lies deep within the brain and constantly integrates sensory input from the outside world. It is connected to cognitive areas such as the cortex and the hippocampus, but also to distinct areas in the brainstem like the locus coeruleus, which is the main noradrenergic nucleus in the brain and regulates stress and panic responses. Directly below the thalamus and as such also closely connected to this ‚awareness hub‘ lies the hypothalamus.
The hypothalamus is a very complex brain area with many different functions and nuclei. Some of them are involved in the control of our circadian rhythm and internal clock – the deciphering of which was awarded the 2017 Nobel Prize in Physiology or Medicine. But the main task of the hypothalamus is to connect the brain with the endocrine system (i.e. hormones) of the rest of the body. Hormones like ghrelin, leptin, or insulin are constantly signalling your brain whether you are hungry or not. They do so via several direct and indirect avenues, such as blood sugar levels, monitoring energy storage in adipose cells, or by secretion from the gastrointestinal mucosa.
There are also a number of mechanosensitive receptors that detect when your stomach walls distend, and you have eaten enough. However, similarly to the hormonal signals, the downstream effects of these receptors also take a little while to reach the brain and be (consciously) noticeable. Thus, the slower you eat, the less likely you will be to over-eat, because the satiety signals from hunger-hormones and stomach-wall-detectors will reach your consciousness only after about 20 to 30 minutes.
Leaving the gut and coming back to the brain, the hypothalamus receives endocrine and neuropeptidergic inputs related to energy metabolism and whether the body requires more food. Like most brain structures, the hypothalamus is made up of several sub-nuclei that differ in cell-type and downstream-function. One of these nuclei, the arcuate nucleus of the hypothalamus, is considered the main hub for feeding and appetite control. Within it there are a number of signalling avenues that converge and that – if altered or silenced – can induce for instance starvation. Major signalling molecules are the Neuropeptide Y, the main inhibitory neurotransmitter GABA, and the peptide hormone melanocortin. The neurons in the arcuate nucleus are stimulated by these and other signalling molecules in order to maintain energy homeostasis for the entire organism. There are two major subclasses of neurons in the arcuate nucleus that are essential for this homeostasis and that, once stimulated, cause very different responses: activation of the so-called POMC neurons decreases food intake, while the stimulation of AGRP neurons increases food intake. And this circuit even works the other way around: researchers found that by directly infusing nutrients into the stomach of mice, they were able to inhibit AGRP neurons and their promotion of food intake.
Given this intricate interplay between different signalling routes, molecules, and areas it is not surprising then that a disrupted balance between all of these players could be detrimental. Recent studies identified one key player that can either keep the balance or wreak havoc: the gut microbiome.
Bacteria colonising intestinal villi make up the gut microbiome. Picture/Credit: ChrisChrisW/iStock.com
The gut microbiome is the entirety of the microorganisms living in our gastrointestinal tract, and they can modulate the gut-brain axis. Most of the microorganisms living on and within us are harmless and in fact are very useful when it comes to digesting our food. However, sometimes this mutually beneficial symbiosis goes awry, and the microbes start ‚acting out‘. For instance, they can alter satiety signals by modulating the ghrelin production and subsequently induce hunger before the stomach is empty, which could foster obesity. They can also block the absorption of vital nutrients by taking them up themselves and thereby inducing malnutrition. A new study which was published only last month revealed that Alzheimer patients display a different and less diverse microbiome composition than healthy control subjects. Another study from Sweden even demonstrated that the specific microbiome composition occurring in Alzheimer’s patients induces the development of disease-specific amyloid-beta plaques, thereby establishing a direct functional link between the gut microbiome and Alzheimer’s disease – at least in mice. Similarly, the composition and function of the microbiome might also directly affect movement impairments in Parkinson’s disease. In addition, there is also mounting evidence that neuropsychiatric diseases such as anxiety or autism are functionally linked to the microbiome.
Moreover, even systemic diseases such as lung, kidney and bladder cancers have been recently linked to the gut microbiome. Albeit, in this case, not the disease development and progression seem to be directly related to our gut inhabitants. Instead, the researchers found that if the microbiome of the cancer patients was disrupted by a recent dose of antibiotics, they were less likely to respond well to the cancer treatment and their long-term survival was significantly diminished. It seems that the treatment with antibiotics disrupts specific components of the microbiome, which then negatively affects the function of the entire composition.
While the cause or consequence mechanisms between these different afflictions and an altered microbiome have not been solved yet, it seems certain that it is involved in more than digestion. Hence, the already intricate gut-brain axis is further complicated by the gut microbiome, which not only affects when and what we eat, but can also determine our fate in health and disease.