Published 19 December 2019 by Arunima Roy
Sins and Ghosts: Epigenetics as a Hot Research Topic
In the far north of Sweden, in Norbotten county, lies a charming locality, Överkalix. Situated close to the scenic river Kalix and with just under a thousand residents, Överkalix harbours some truly breathtaking vistas. Here, meticulous records maintained by civic and religious authorities since the 19th century give a fascinating glimpse into life in the 1800s. These records tell of times when famines struck and of times when harvests were plentiful. This idyllic setting with its trove of historical data has been a game changer in our understanding of human health and its environmental determinants.

Researchers discovered a startling pattern in the descendants of 19th century Överkalix residents. A paternal grandfather’s caloric intake was related to longevity and mortality in grandsons, not granddaughters. A paternal grandmother’s food intake was linked to cardiovascular risk and mortality in granddaughters, but not grandsons. Such effects were only seen if ancestors’ caloric intake deviated during specific growth periods in their childhoods.
This work led to two insights: one, that it was possible to transmit changes in response to the environment down to successive generations. Two, the health effects of one’s ancestor’s environment could be sex-dependent. These effects were suggested to be ‘epigenetically’ regulated.
Immediately, there was skepticism. Transmission of environmental effects from grandpa and grandma to grandkids sounded hocus-pocus. Criticisms of the study also included modest sample sizes and issues with multiple testing. It did not help that sensational taglines like ‘sins of the father’ abounded online. However, other studies emerged that started showing comparable results (see for example Vågerö et al.).
Then in 2014, Rachel Yehuda’s group demonstrated altered epigenetic patterns in children of Holocaust survivors. Their work revealed an epigenetic change associated with the glucocorticoid receptor, and an alteration in cortisol (a stress hormone) secretion, in children whose parents had post-traumatic stress disorder
No scientist publishes without a full disclosure and so here’s mine: since 2017, I have been working in epigenetics. So far, my conversations with those not in this field has been on the lines of “You work in epigenetics? Woah! That’s a hot field to be in now.”
I partly see where they are coming from. Epigenetics presents an uncharted territory and believe you me, I too have salivated at the thought of all the discoveries waiting to be made. However, having seen the field up close, my enthusiasm has tempered, not because there aren’t any promising leads, but because there seem to be too many of them. The closer you look at it, the more complex and nefariously murky the picture becomes.
At its core, epigenetics are changes that alter the way genes function. That is, genes provide the basic template; all the instructions that our body’s cells need to survive and to function. However, they don’t all continuously work and keep the machinery chuffing away, no. Genes can be, and are, turned on or off. It is here that epigenetics makes its debut. Epigenetic patterns decide which gene is turned on and when. Subsequently, epigenetics controls what instructions reach a cell, controlling the cell’s actions and functions.
The word ‘epigenetics’ may come across as vague. It literally means “over, outside, or around” the genes. That doesn’t say much about what these nebulous forms are and where they decide to park themselves other than in the general, and very obvious, vicinity of genes.
Epigenetics is, in fact, a broad term that encompasses (m)any alterations that induce genetic switching-ons-and-offs without changing the actual genetic template. And here’s where the complexity begins, innocuous at first, and then bewildering. For example, the most commonly studied form of epigenetic change involves attachment of methyl moieties to specific sites on the genome. Current technological capabilities allow us to examine approximately 850,000 sites on the genome where methylation can occur. It can be difficult, to say the least, to make sense of simultaneous changes at multiple sites. But 850,000 are not all. These are select locations based on our current understanding of “biologically relevant” methylation marks. In fact, methylation changes can potentially occur at close to 30 million sites on our genome. Neither do we have the capacity to examine all 30 million locations nor is it very apparent how we could interpret all that information. Needless to say that the function of most of these methylation marks is unknown.
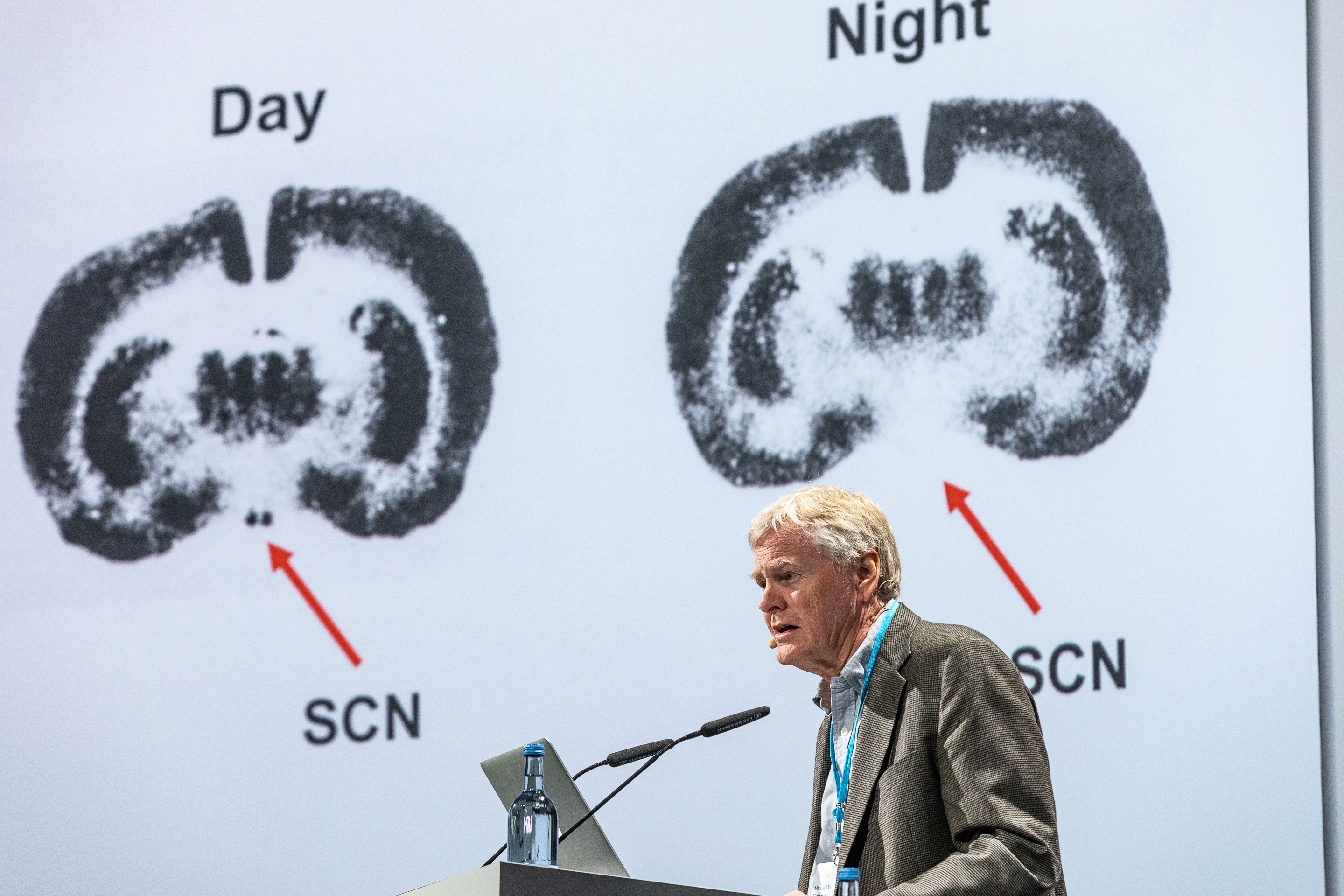
Of course, other epigenetic changes can occur too. Changes to the manner in which DNA is packaged can alter which gene is active and which not. Further alterations can occur downstream, after the gene has produced a set of instructions for the cell: the process of carrying and fine-tuning the message can induce changes. It is not one single epigenetic change, either methylation, or packaging, or messenger induced alteration, that modifies the genetic message. Rather, multiple epigenetic changes, at disparate locations, could be engaged in a tussle to decide if the genetic switch goes on or off. Add to this menagerie the fickle nature of epigenes: epigenetic changes can be induced by many environmental factors. Epigenetic changes can occur on timescales of decades, years, hours and minutes. Epigenetic changes can show circadian rhythmicity, the internal process that regulates our sleep-wake cycle and that was discovered by the 2017 Nobel Laureates in Medicine Jeffrey C. Hall, Michael Rosbash and Michael W. Young.
These extant possibilities give a glimpse into the impressive mechanisms that control cellular function and the intricate precision this machinery is capable of achieving. Without mincing words though, it will be a nightmare to fully understand the epigenetic regulatory mechanism.
Nevertheless, the intrepid have made substantial headway and produced thrilling results. Take for example the discovery of the epigenetic clock: in 2013, Steve Horvath demonstrated that age can be accurately estimated from epigenetic changes. As of 2019, this work was extended to understand if longevity could be improved. And just a few weeks prior to writing this post, an article was published that estimated dogs’ (Labrador retrievers) age in human years, based on epigenetics.
Some years ago, I gave a talk on the epigenetic clock. This predated my own entry into epigenetic research so my understanding, and hence my talk, could be half-baked and superficial (that is, I implore you not to google it). It was around this time, hooked by epigenetics, that I chanced on the Nova PBS documentary ‘ghost in your genes’. Dramatic name and ominous music aside, the documentary gives a good overview to the subject. With a 4 star ranking on IMDB and a 5-star ranking on Amazon, the documentary shows the appeal of epigenetics. Epigenetics, despite its exasperating complexities, has much to offer in terms of ground-breaking results and henceforth hope.
To close the circle, can epigenetic changes be transmitted down several generations as suggested in the Överkalix study? Maybe or maybe not. Epigenetic marks, particularly methylation marks, were thought to be completely removed as an embryo is formed, meaning that either parent’s epigenetic patterns would not get passed on to the baby. But recent work shows that this is not always the case. So the jury is out, scientifically jousting. Can such changes be passed down not just two or three, but hundreds of generations? I hope not. Humankind has had a penchant for war and the possibility of us all carrying suboptimal epigenetic marks due to millennia of stressful experiences seems both unappeasing and far-fetched. The foregone conclusion, however, is that more work needs to be done to understand the complexities and nuances of epigenetics. The field is a goldmine awaiting prospectors as interesting questions remain to be examined. If you are intrigued, adventurous explorers are needed here.